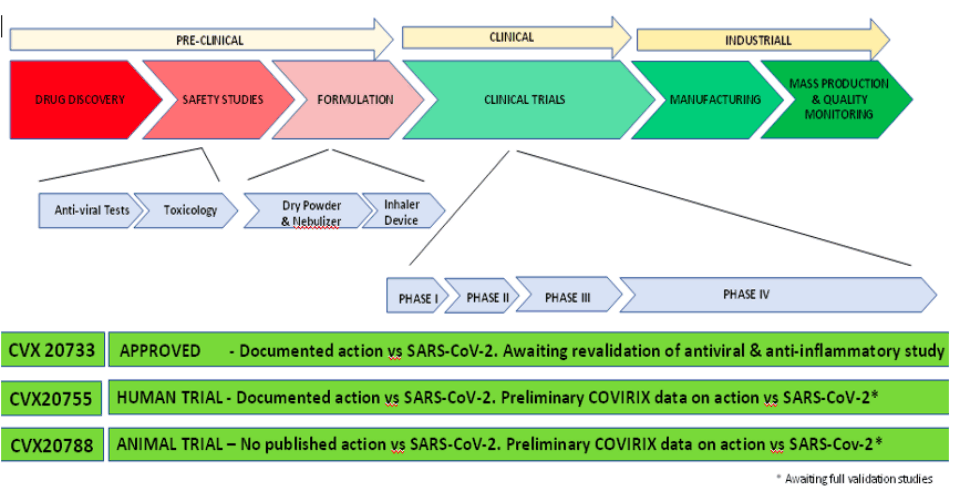

Initiation of Clinical Trials for Lead Compound CVX 20733
Q3 – Q4 2023 Submission for Regulatory and Ethics approval for studies in Australia, Nepal & India
Q3 2023 – Q3 2024 With existing systemic toxicology data
Chemistry, manufacturing and controls. Incudes micronisation and dry powder formulation
Research grade and demo batches through to GMP scale up, to formulation transfer and and cGMP product manufacturing
Clinical Phase I 16 – 24 Healthy volunteers
Q4 2023 – Q1 2024 Irritability, Tolerability, Safety, and Pharmacokinetic studies
Clinical Phase II 200 – 250 patients
Q1 – Q2 2024 Proof of concept. Dose, Scheduling and Efficacy.
Clinical Phase III 1500 - 3000 patients
Q3 2024 onwards Multi-centre, multi-national centres
Parallel Drug Development R & D on Follow-on compounds to explore Q3 2023 onwards
Compound class efficacy against other viral (RNA – enveloped) viral respiratory diseases eg. (influenza, parainfluenza, respiratory syncytial virus), as well as systemic diseases like Dengue and Ebola.
Full animal studies for primary screening, animal efficacy models, and comprehensive animal safety with ADME (adsorption, distribution, metabolism, excretion)
Collaborative studies with the Blumberg Institute in Pennsylvania, USA to explore the efficacy of the COVIRIX compound class in (i) Reducing the mutation rate of SARS-CoV 2 virus (ii) define any added synergy in therapeutic benefit of combining COVIRIX compound class with other available antivirals (iii) develop novel pro-drugs of the COVIRIX compound class to develop additional oral equivalents without toxicity.
Development of a re-purposed non-steroidal anti-inflammatory agent for respiratory diseases (eg. exacerbations of asthma, COPD and Interstitial Lung Disease)